Great Info About How To Learn Electron Configuration

Electron configurations describe where electrons are located around the nucleus of an atom.
How to learn electron configuration. There are electrons in all three p sublevels instead of. First you should write their normal electron configuration and then when you remove electrons you have to take them from the outermost shell. The rule to calculate the number of electrons that each shell can hold is 2n 2.
Start writing electron configuration from the very first element (i.e., hydrogen) all the way up to sulfur. For example, the electron configuration. It is written out, as opposed to orbital diagrams which are depicted pictorially.
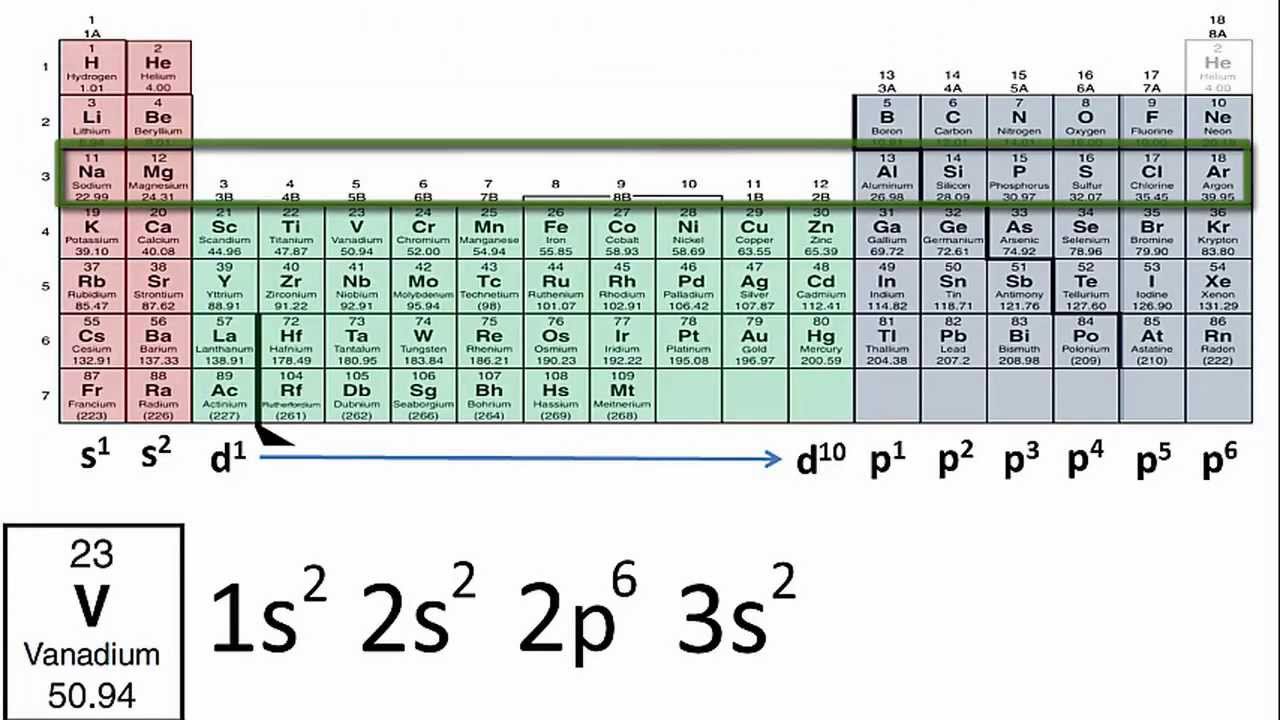
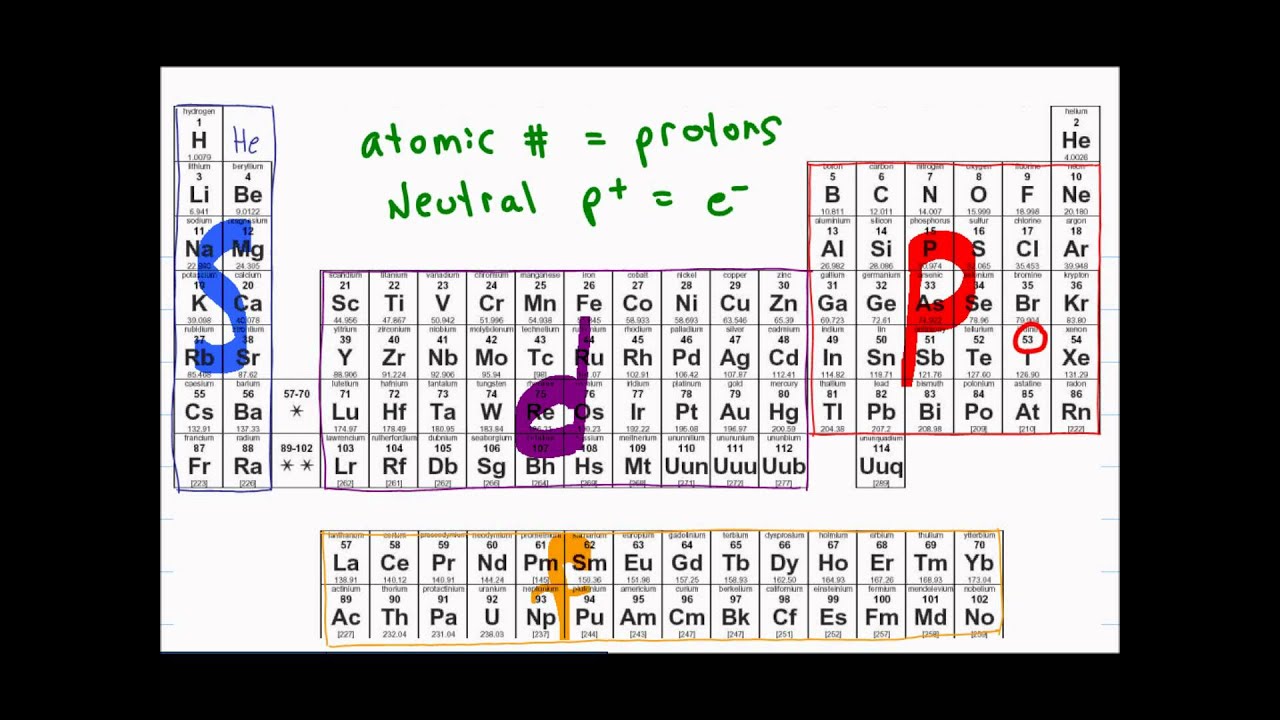
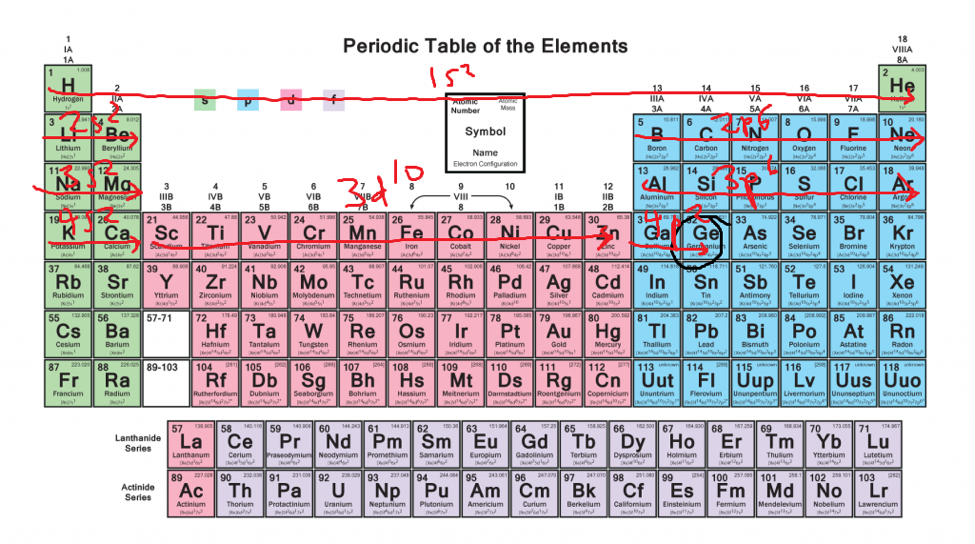
#protons=#electrons so if you have, let's say, 9 protons (fluorine). 119 rows shorthand electron configuration full electron configuration electron shell arrangement; The main electron shells are numbered 1 to 7.
Look at the box and arrow diagram for oxygen. It contains plenty of practice problems and examples including the. An up arrow and a down arrow have different “spins.”.
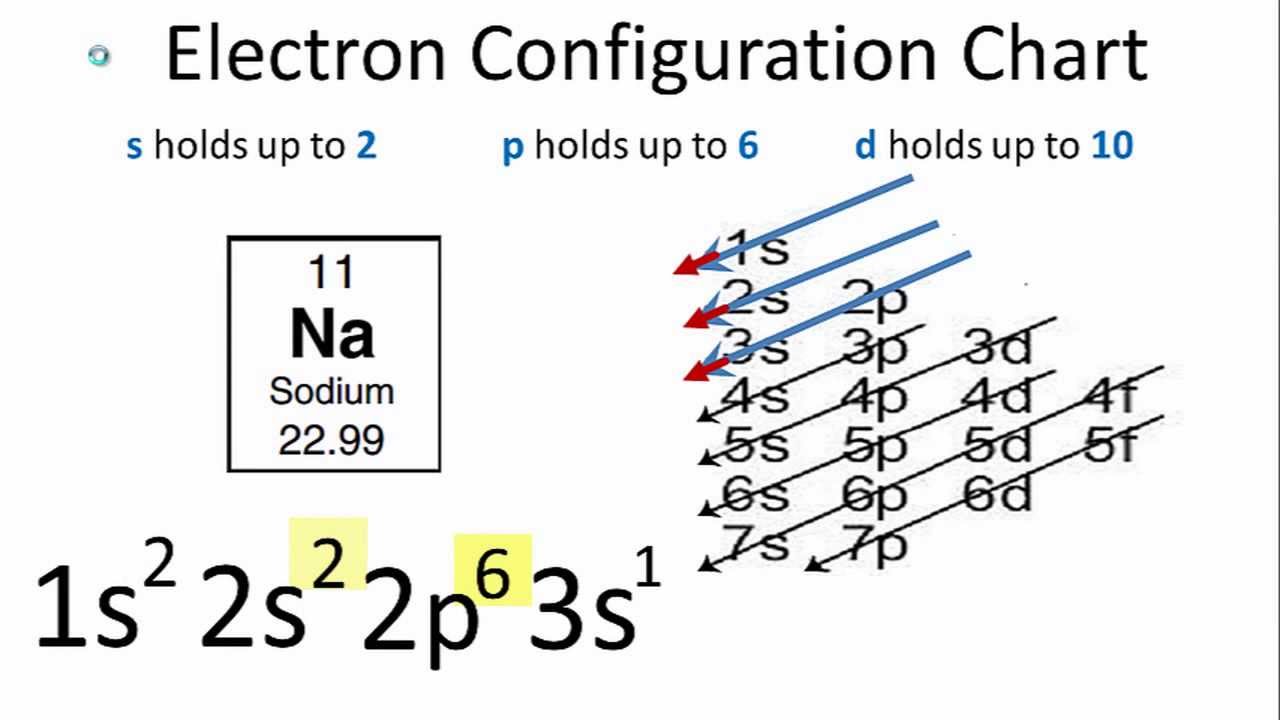
Each subshell has one or more. Electron configuration of hydrogen (h) 1s 1: So the electron configuration of sulfur will be 1s 2 2s 2 2p 6 3s 2 3p 4.
The first shell is 2 (1) 2 which gives you 2 electrons. Note that this is not always the same way. Using these rules, the electron configuration of any element can be theorized.
This chemistry video tutorial provides a basic introduction into electron configuration. Each shell has one or more subshells within it. The boxes and arrows method uses boxes to show orbitals and arrows to signify electrons.
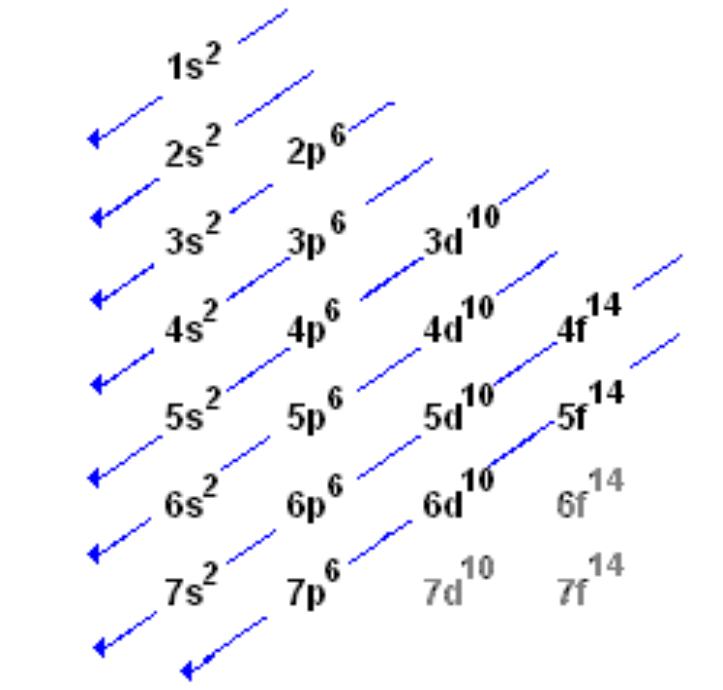
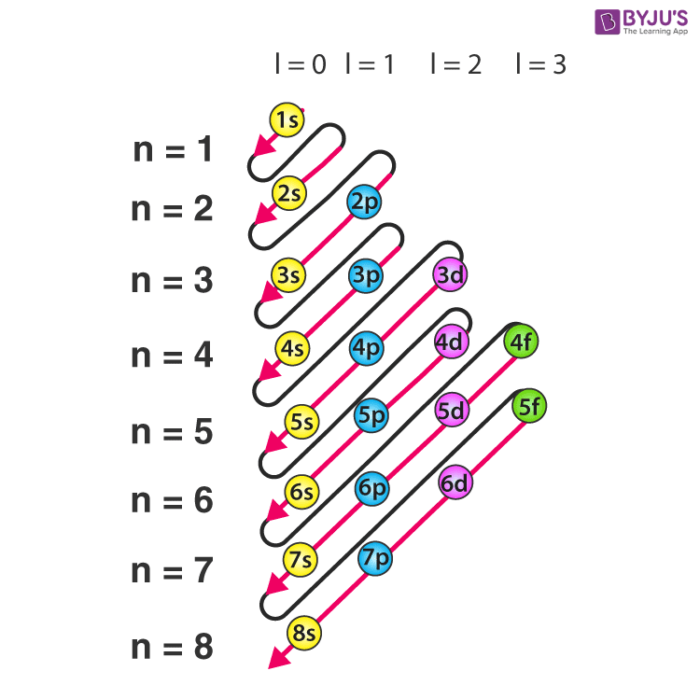
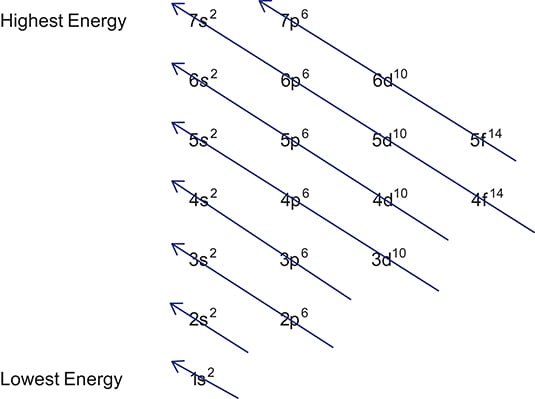
In the diagram above the energy levels are. Electron configuration is shorthand for the arrangement of electrons in atomic orbitals.